Golden Biotechnology Corp.(GoldenBiotech, 4132.TWO), a leading Taiwanese biopharmaceutical company, announces that its new drug Antroquinonol (HOCENA) outperforms the other listing drugs for the treatment of relapsed AML (acute myeloid leukemia) patients in its Phase 2 clinical study conducted in Russia. The outcome measures demonstrated higher remission rates (50%, CR/Cri) and survival rates guaranteeing fewer patients will require blood transfusions. This has made high safety possible during breakthroughs for AML treatment with remarkable monotherapy without combined chemotherapy. The new drug Antroquinonol has been granted orphan drug designation (ODD) by the U.S. FDA for the treatment of AML in 2015. GoldenBiotech has achieved excellent breakthrough in the unmet medical needs in AML therapy which many global pharmaceutical companies are targeting.
Clinical Results Summary:
*Primary Outcome Measure- overall remission rate (CR/CRi): 50%
*Secondary Outcome Measures
80% of patients showed that their blasts count were less than 5% 90% of patients did not need blood transfusion during trial period 100% of patients were all alive during trial period (6 months) No serious side effectThe primary efficacy indicator results exhibit that the overall remission rate (CR/CRi) of patients with relapsed acute myeloid leukemia reaches 50%. This is higher than those of other listing drugs in their Phase 2 trial ranging approximately from 21%~33% in (CR/CRi). 80% of patients' abnormal blasts counts in bone marrow or blood were decreased to be less than 5% after treatment. For the secondary outcome measures results the overall survival rate of patients with relapsed AML reached 100% by the 6th month after treatment. This amplifies the possibility of patients receiving bone marrow transplantation. Furthermore, 90% of patients do not need to undergo blood transfusion during treatment with Antroquinonol (Hocena). Oral treatments and the decrease in blood transfusions have greatly improved the quality of life of AML patients. There is no significant side effect (SAE) that has occurred during the 6-month treatment period. The dose trial is administered orally at home by taking 200 mg twice a day.
The trial of Antroquinonol (HOCENA), Protocol No. GHAML-2-001, was an open-label, non-randomized, Phase 2a study to evaluate efficacy and safety/tolerability profiles in adult patients with relapsed acute myeloid leukemia (AML) or at initial diagnosis when no intensive treatment is possible.
There is no standard treatment for relapsed/refractory adult acute myeloid leukemia (AML) patients. Based on the confidence achieved in the trial results for the treatment of second-line and recurrent AML patients, GoldenBiotech will apply the clinical trial (IND) to the U.S. FDA for the Phase 2 clinical trials as the first-line treatment of AML and continue to expand the spectrum of indications of Antroquinonol in other leukemia treatment fields including multiple myeloma (MM), chronic myelogenous leukemia (CML), and pediatric leukemia.
The majority of AML patients need to undergo blood transfusions and chemotherapy to kill cancer cells due to abnormal differentiation of blood cells and proliferation of myeloblasts. Current therapies are combined with chemotherapy often causing serious side effects resulting in poor prognosis of treatment for relapsed AML patients. AML ranks the second highest incidence in all leukemia but it has the highest mortality rate and the lowest five-year survival rate (about only 25%).
It is estimated that there will be about 62,000 patients with acute myeloid leukemia (AML) in 2020. According to the reports from GlobalData and other institutions, the global AML therapeutic drug market is expected to increase from USD 1.4 billion in 2019 to 5.1 billion in 2029 with a compound annual growth rate (CAGR) of 13.6%.
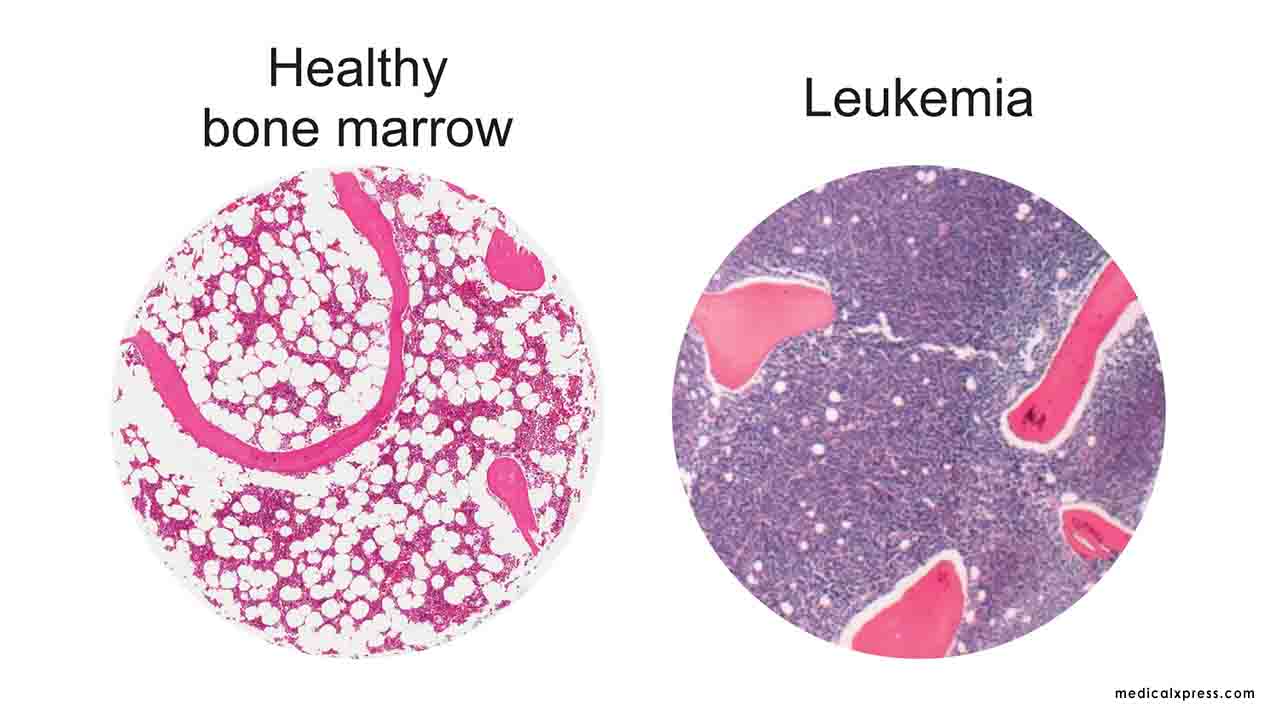
 Outperforms listing drugs in relapsed Acute Myeloid Leukemia
Outperforms listing drugs in relapsed Acute Myeloid Leukemia
















.jpeg)

.jpeg)










.jpg)




.jpg)

