Rapid antigen tests may be used to detect Covid-19 patients within the first five days of the illness that will help in quick identification, isolation and treatment of the deadly disease, a study published in the Indian Journal of Medical Research (IJMR) has stated.
The study showed that patients showing positive results in rapid antigen test need to be immediately triaged and those with negative tests should be reconfirmed by a real-time PCR test (RT-PCR).
The study was conducted at the Covid-19 screening and testing outpatient department at the AIIMS in New Delhi between May 31 and July 24 covering 330 people, news agency PTI reported.
The most commonly presented symptoms among the screened participants were fever (31.5 per cent) followed by cough (25.4 per cent), fatigue and malaise (11.8 per cent), headache (3.3 per cent) and runny nose (3.3 per cent). Fifty-seven participants presented with the complaint of a sore throat but only two of them (3.5 per cent) had Covid-19.
Overall, among the positive test results, rapid antigen test detected 63 true positives (19.0 per cent) and gave one false-positive result with respect to the reference standard. Among negative test results, 252 (76.3 per cent) were true negatives and 14 (4.2 per cent) were false negatives.
The overall sensitivity and specificity of the test were 81.8 and 99.6 per cent, respectively, and the test accuracy was 95.4 per cent.
"In conclusion, the rapid antigen test showed an excellent specificity to ''rule-in'' COVID-19 patients within the first five days of illness and had a moderate sensitivity.
Therefore, patients showing positive results need to be immediately triaged and those with negative tests should be reconfirmed by an rRT-PCR," the study stated.
The real-time reverse transcription-polymerase chain reaction (rRT-PCR) test received emergency use authorisation from the Centers for Disease Control and Prevention (CDC) for the qualitative detection of SARS-CoV-2 nucleic acid from the respiratory specimens.
Real-time RT-PCR is the gold standard test for detecting cases of COVID-19. The test requires specialized laboratory setup with specific biosafety and biosecurity precautions to be followed.
It takes a minimum of eight to ten hours from the collection of the swab to reporting of results, which can further increase in resource-limited and high-burden settings, stated the study.
The study, however, mentioned that the advantage of this platform lies in its accuracy of detection as well as the ability to run up to 90 samples in a single run.
The need of the hour is to rapidly detect and isolate positive cases to contain the disease spread, to quickly triage patients with severe acute respiratory illness in emergency departments and to ramp up testing facilities, it said.
About rapid-antigen testing, it said these diagnostic kits and devices facilitate point-of-care testing. These sample kits are based on either antibody detection from blood, plasma or serum or the detection of SARS-CoV-2 antigens from respiratory samples.
Asserting that a test is a promising tool for quick diagnosis of SARS-CoV-2 in field settings, the study mentioned, "There is very limited data on the performance and potential diagnostic utility of a rapid chromatographic immunoassay-based test for SARS-CoV-2 in suspected patients."
Story Source: PTI
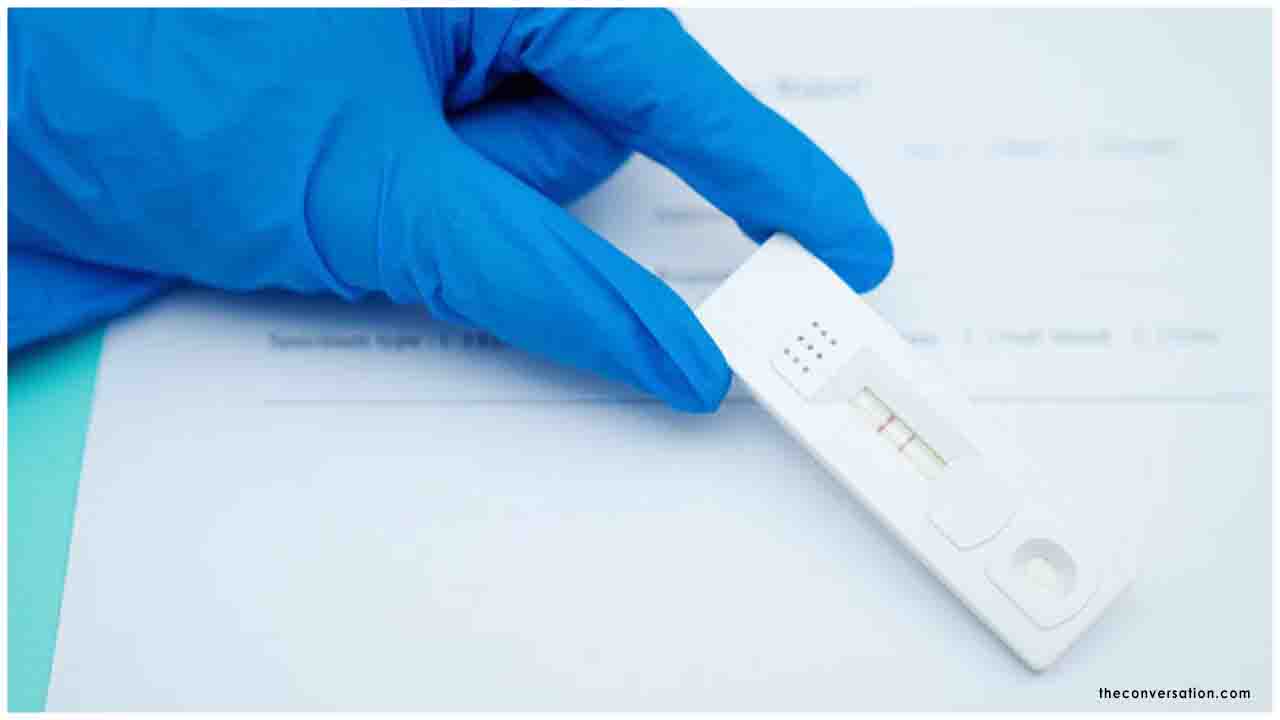
 The advantage of this platform lies in its accuracy of detection as well as the ability to run up to 90 samples in a single run
The advantage of this platform lies in its accuracy of detection as well as the ability to run up to 90 samples in a single run










.jpeg)

.jpeg)
.jpeg)

.jpeg)

.jpeg)


.jpeg)
.jpeg)
.jpeg)


.jpg)


.jpeg)
.jpeg)